The EUA for this product is in effect for the duration of the COVID-19 EUA declaration justifying emergency use of the product, unless the declaration is terminated or the authorization is revoked sooner.įor more information on the EUA authorized uses of the vaccine, refer to the Fact Sheet for Healthcare Providers Administering Vaccine. Those who received the Moderna/Spikevax vaccine when they were below 5 years will continue to receive the same vaccine and dosage their second dose, even if. Those who received the dosage for persons aged 5 to 11 years for their first dose will continue to receive the same dosage for their second dose, even if they. When youre fully vaccinated 14 days from receiving the second dose of either Pfizer or Moderna or the one-dose vaccine from Johnson & Johnson youll be. 12, 2022, the new COVID-19 booster recommendations for people ages 5 years and older is to receive 1 bivalent mRNA booster after completion of a. 
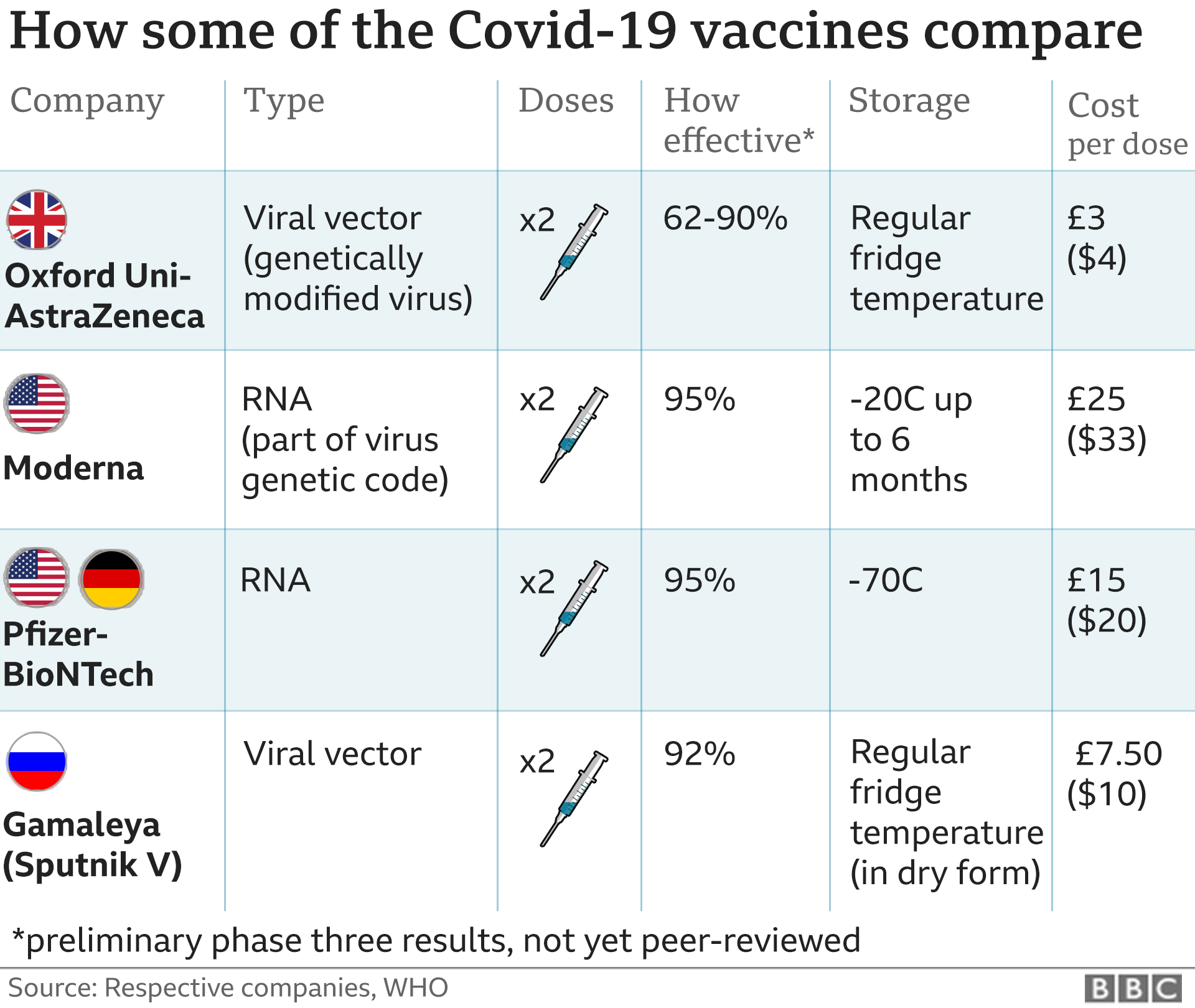
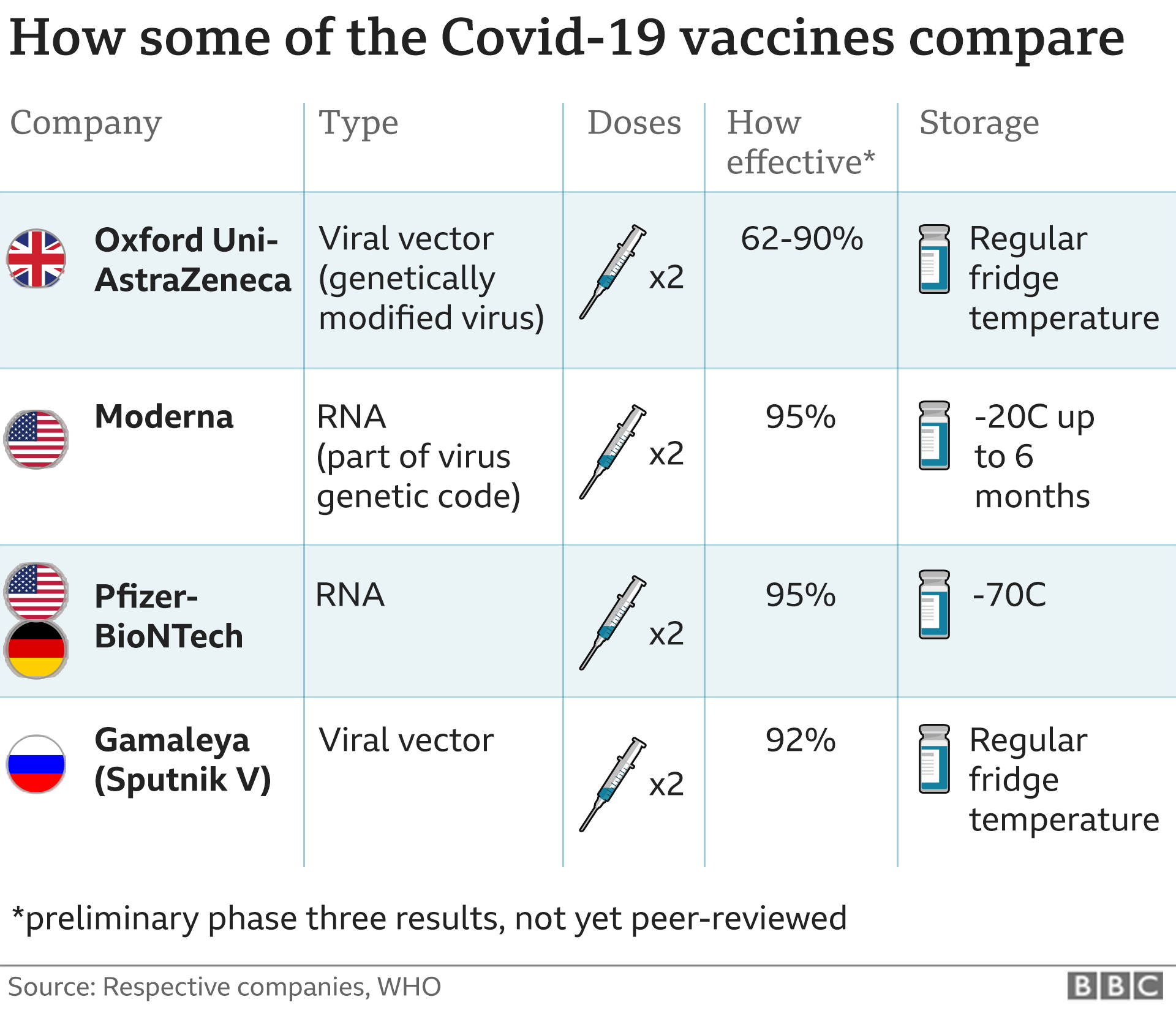
The move came a day after the Food and Drug. Certain additional uses are authorized for immunocompromised patients and patients 65 years and older. The Centers for Disease Control and Prevention has signed off on vaccines for Americans younger than 5 years old, allowing shots to begin immediately. Moderna ’s Covid-19 vaccine was more than 90 effective at protecting against Covid and more than 95 effective against severe disease up to six months after the second dose, the company. Moderna COVID-19 Vaccine, Bivalent is authorized for use in individuals 6 years of age and older who were previously unvaccinated or vaccinated with one or more doses of an approved or authorized monovalent COVID-19 vaccine at least 2 months after receipt of any monovalent COVID-19 vaccine. However, people who are at greater risk of severe illness from COVID-19, such as those with weak immune. Moderna COVID-19 Vaccine, Bivalent is authorized for use in individuals 6 months through 5 years of age who were previously unvaccinated or vaccinated with one or two doses of Moderna COVID-19 Vaccine (no longer authorized). Most people need 2 first doses of a COVID-19 vaccine. People aged 65 years and older or people more likely to get very sick from COVID-19 should get the 2nd dose of Novavax COVID-19 vaccine 3 weeks after the 1st. Moderna COVID-19 Vaccine, Bivalent has not been approved or licensed by the FDA, but has been authorized by the FDA, under an Emergency Use Authorization (EUA) to prevent Coronavirus Disease 2019 (COVID-19).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
